Children Aged 5 to 15 Injected with COVID-19 Shots, Will 1 Million Babies be Injured and Killed

by Brian Shilhavy
Editor, Health Impact News Source
The FDA and CDC will consider injecting babies and infants between the ages of 6 months and 5 years old with COVID-19 vaccines from Pfizer and Moderna in the coming week.
In May of 2021 the FDA and CDC authorized Pfizer COVID-19 vaccines for children between the ages of 12 and 15, and at the end of October in 2021 they authorized Pfizer COVID-19 vaccines for children between the ages of 5 and 11.
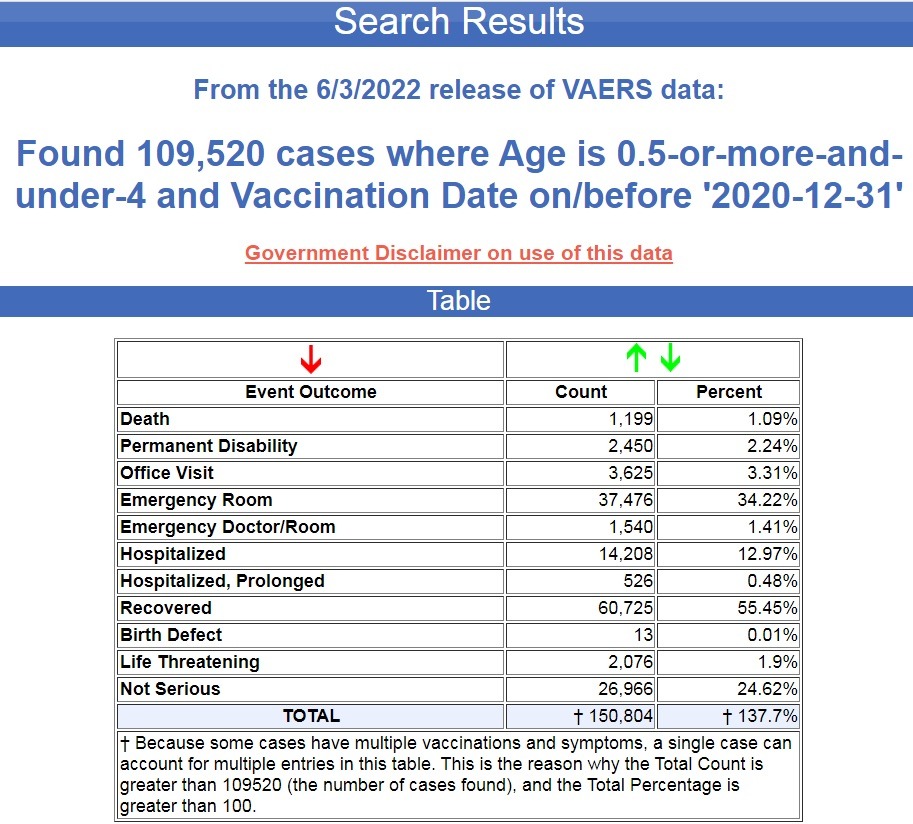
So I am going to publish Government data on cases of adverse reactions that have been filed with the Vaccine Adverse Events Reporting System (VAERS) for these age groups subsequent to the authorization for these vaccines for these age groups, something that neither the government nor the pharma-funded corporate media will publish, and yet can be verified by anyone with Internet access since this is public knowledge.
In this report today, I will also include cases of fetal deaths recorded in VAERS after their mothers were injected with COVID-19 vaccines, as 2nd and 3rd trimester unborn children are closer in age to 6-month-old babies that the FDA and CDC will soon decide to start injecting with these shots.
Anyone who plans to be present next week in these advisory committee meetings is free to copy and use this data.
Children Between the Ages of 12 and 15 Starting in May 2021 (12 Months Data)
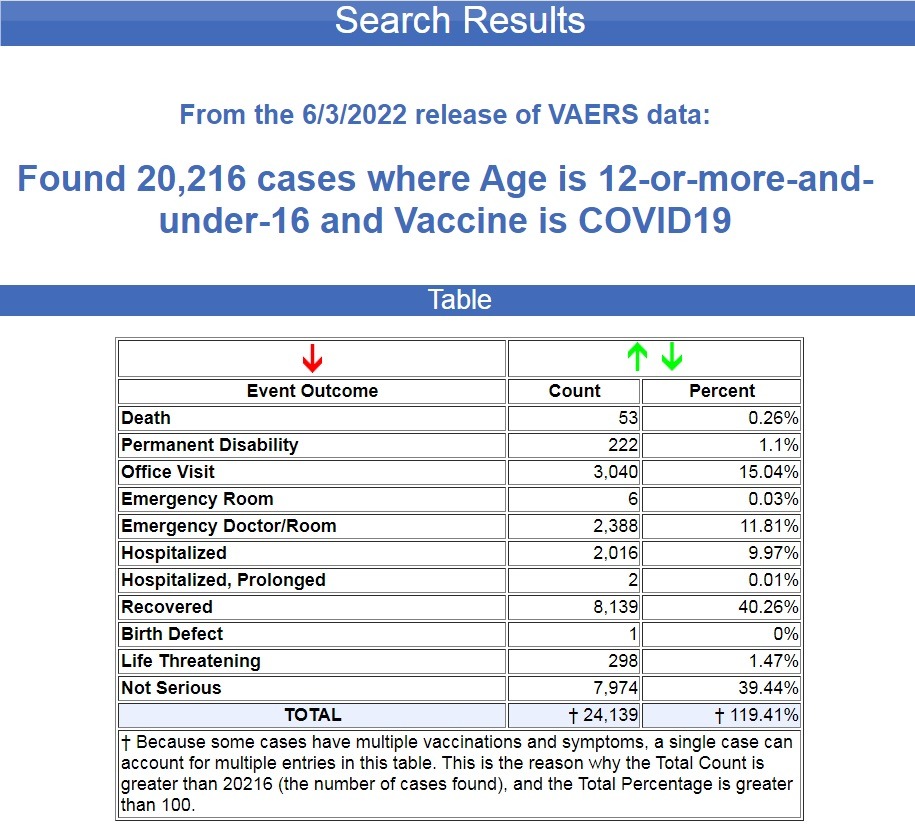
In the 12 months following the FDA and CDC authorizing the COVID-19 Pfizer vaccines for children between the ages of 12 and 15, 20,216 cases of injuries and deaths were filed in VAERS.
That averages out to 1,685 cases per month, and let’s not forget that VAERS is seriously under-reported, and the under-reported factor is 41x. (Source.)
For the previous 30 years (360 months) before the COVID-19 vaccines were authorized, there were 39,041 cases of deaths and injuries filed in VAERS following all FDA-approved vaccines for this age group, an average of 108 cases per month. (Source.)
That’s a 1,460% increase in adverse events for this age group following the COVID-19 shots.
Children Between the Ages of 5 and 11 Starting at the End of October 2021 (7 Months of Data)
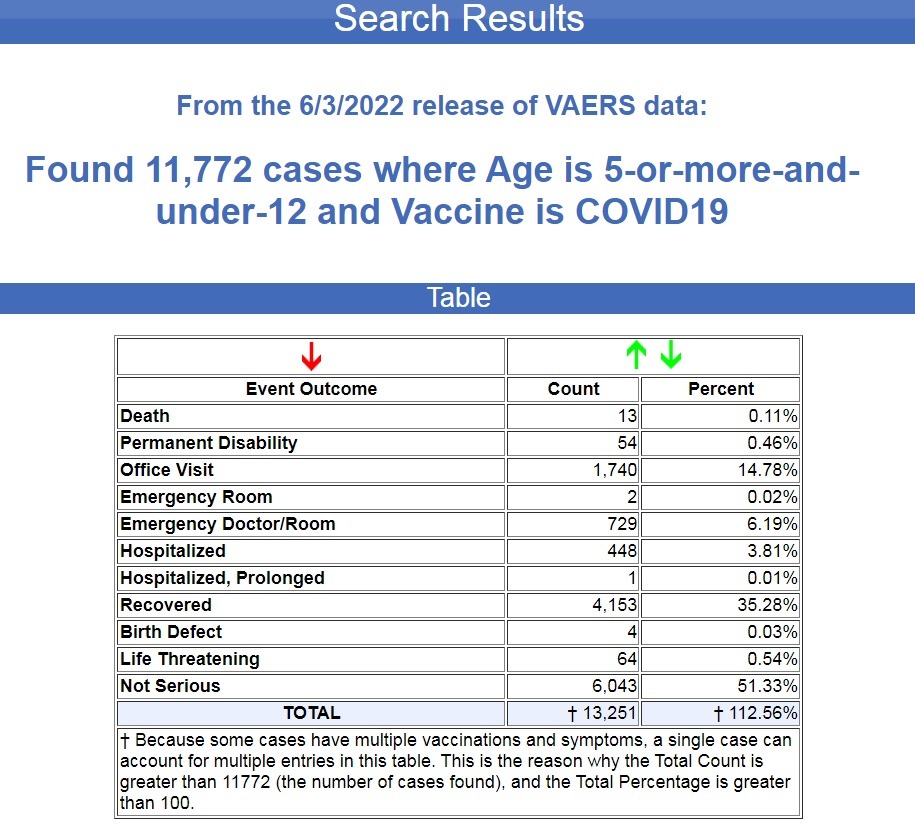
In the 7 months following the FDA and CDC authorizing the COVID-19 Pfizer vaccines for children between the ages of 5 and 11, 11,772 cases of injuries and deaths were filed in VAERS.
That averages out to 1,675 cases per month.
For the previous 30 years (360 months) before the COVID-19 vaccines were authorized, there were 60,952 cases of deaths and injuries filed in VAERS following all FDA-approved vaccines for this age group, an average of 169 cases per month. (Source.)
That’s an 891% increase in adverse events for this age group following the COVID-19 shots.
How Many Deaths and Injuries Can We Expect in Babies and Toddlers Between the Ages of 6 Months through 4 Years Old if the COVID-19 Vaccines are Authorized for this Age Group?
Using this data from the government-run VAERS database, we can now predict how many babies and toddlers will die or become injured following COVID-19 injections, should the FDA and CDC authorize their use for this age group this coming week.
As you can see from the data above, there were significantly more cases filed for FDA-approved vaccines for the past 30 years among the younger age group, between the ages of 5 and 11.
This can probably be accounted for based on the CDC child immunization schedule, found here.
For children between the ages of 11 and 15, there are 4 doses of vaccines (non-COVID) per year recommended.
For children between the ages of 4 and 10, there are 7 doses of vaccines (non-COVID) per year recommended.
But for younger children between birth and 3 years old, there are 28 doses of vaccines (non-COVID) per year recommended.
So for the previous 30 years prior to the introduction of the COVID-19 vaccines, here are the cases filed in VAERS for this age group:
So now we have enough data to predict how many cases might be filed in VAERS for injuries and deaths following COVID-19 injections into babies and toddlers in this age group, which already is being injected with far more vaccines than older children.
For the previous 30 years this age group has averaged 304 cases filed per month for FDA-approved vaccines.
The number of adverse events per month for 5 to 11 year olds, and 12 to 15 year olds, following the EUA COVID-19 vaccines, was very similar, averaging 1680 cases filed per month between the two age groups.
The unknown factor here is how many doses of COVID-19 vaccines were distributed within each of the age groups of children already authorized. It has been widely reported that far fewer parents agreed to the COVID-19 vaccines for the younger age group already authorized, ages 5 to 11, than for the older children above the age of 12, although the number of adverse events per month was about the same.
It is probably safe to estimate that the younger children, with a less developed immune system, suffer adverse events more frequently than older children.
It is also probably safe to assume that even fewer parents will agree to allow their infants and toddlers to receive COVID-19 shots compared to older children.
So if we keep the average of adverse events filed following the existing age groups of children for the EUA COVID-19 shots at 1680 cases per month, we can expect over 10,000 injuries and deaths of babies and toddlers filed in VAERS within the first 6 months following authorization for this age group.
It would be a 453% increase over deaths and injuries recorded after FDA-approved vaccines for the previous 30 years.
And again, this is just a fraction of actual cases that are reported, and if we use the already determined unreported factor in VAERS for COVID-19 shots of 41x, that would result in 410,000 injuries and deaths in this age group within the first 6 months.