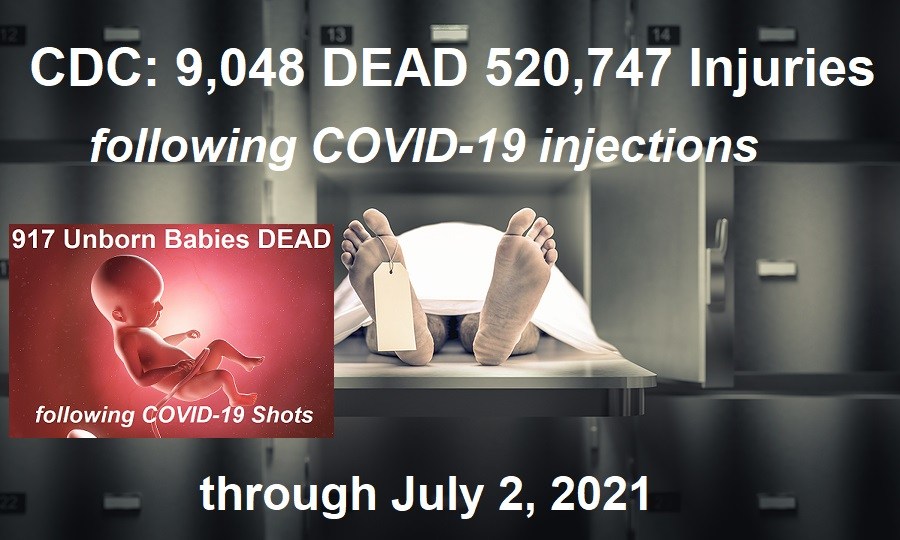
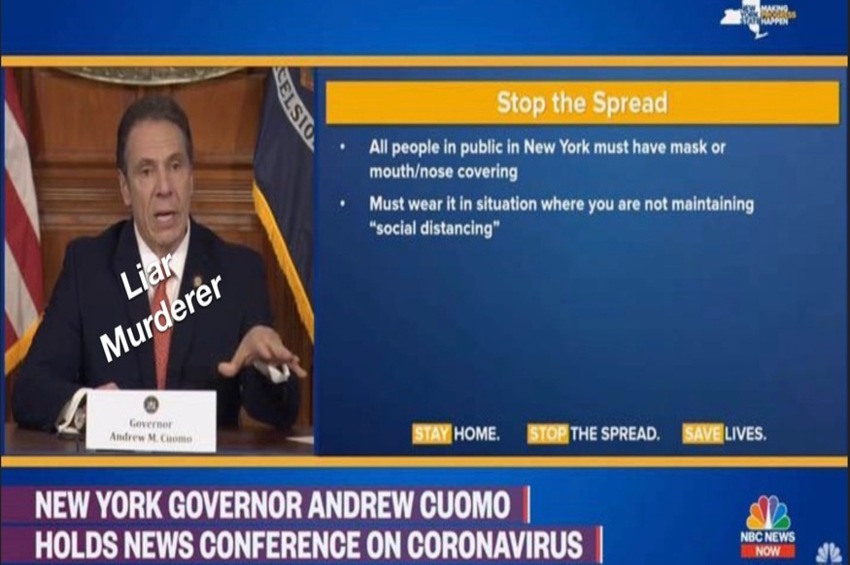
CDC to Withdraw Emergency Use Authorization for RT PCR Test
by Brian Shilhavy Editor, Health Impact News The CDC quietly announced last week that it was withdrawing its request to the FDA for Emergency Use Authorization (EUA) of the 2019-Novel Coronavirus (2019-nCoV) Real-Time RT-PCR Diagnostic Panel, the assay first introduced in February 2020 for detection of SARS-CoV-2. Most of the public is probably unaware that similar […]Read More